Human health & rejuvenation
Development of sustainable healthcare practice for the future

We help withPluripotent stem cell research
From setting up a laboratory & standardised workflows, recruitment & training to experimental design, analysis and publication, with everything in between.
Manufacturing for cell therapy
From designing & operating facilities to translation of stem cell research protocols for compliance with Good Manufacturing Practice and regulatory authority requirements, with everything in between.
Enabling technology
Procurement and validation of state of the art technologies to improve your practices and outputs.
Sustainability
Future proofing your goals for ethics, affordability, scalability, safety, effectiveness and ecology.
OUR EXPERIENCE
Establishing Scotland’s advanced stem cell manufacturing
I founded and served as Chief Scientific Officer of Roslin Cells Ltd from 2005 to 2019. This established Scotland's advanced cell therapy manufacturing capability now operating as Roslin CT. I led the design, installation, operational and performance qualification of quality assured Good Manufacturing Practice compliant facilities. I was responsible for regulatory licencing; ethical procurement of donated tissue; stem cell banking and co-development of Advanced Tissue Medicinal Products (ATMP) supporting partners and clients. The clinical-grade cell lines established continue to be used as ATMP source material.



QR to hPSCReg registry information for clinical grade Roslin Cells human embryo stem cell lines.
Hot start for European Bank for induced pluripotent Stem Cells


From 2017-2019 I led the hot start establishment of a standardise quality-controlled resource of induced pluripotent stem cells from laboratories across Europe for scientific discovery across both academia and industry. This capacity was commercialised in Censo Biotechnologies that now operates as Axol Bioscience.
Innovating pluripotent stem cell based therapy and discovery
From 2020 to 2025 I founded and executively directed Stroma Therapeutics Ltd, innovating human pluripotent stem cell derived mesenchymal stromal cells for therapy and as a discovery engine for tissue reparative and immune modulating factors. Proprietary know-how and data including cell RNAseq and secretome proteomic data are available for licensing.
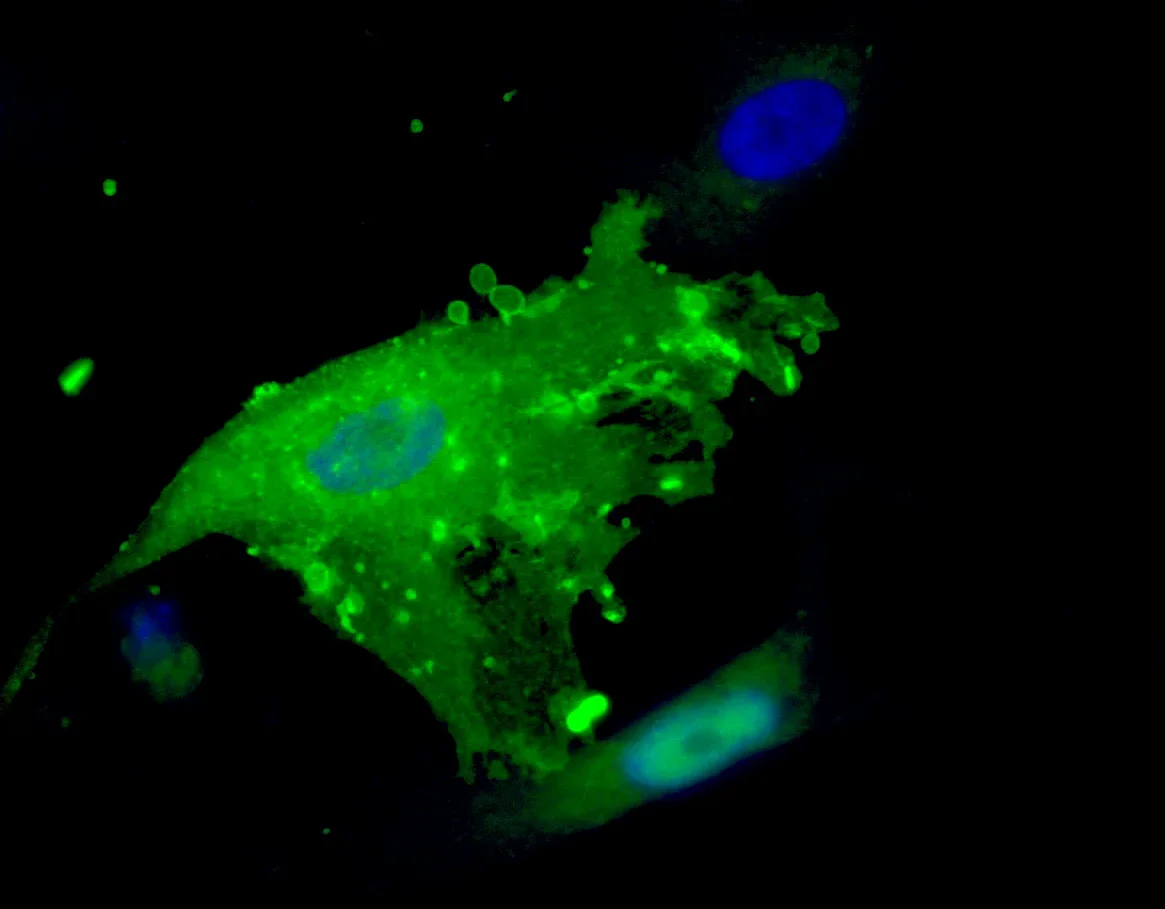
Human pluripotent stem cell derived mesenchymal stromal cells (MSC) immunostained for CD73 (Ecto-5´-nucleotidase), a biomarker of MSC identity., in the process of cytoplasmic blebbing inducible by apoptosis, programmed cell death..
Building for sustainability