Stem Cell 101
Survey of current application domains: disease modelling, cell therapy, species preservation, agri-food, reproduction. Where each stands and what limits progress.
What GMP means in the context of stem cell products. How Quality by Design differs from post-hoc testing. Practical implications for anyone building or using tools in clinical pipelines.
What assays and markers are used to assess stem cell identity and quality. Where current tools fall short: epigenetic stability, functional potency, and subtle state shifts.
A practical overview of core laboratory and manufacturing methods used in stem cell science. What each does, why it matters, and where limitations lie.
How cells can be reprogrammed. Covers SCNT (cloning), iPSC generation, and direct reprogramming. Grounds the concept in the history from Dolly onward.
An accessible overview of major stem cell categories, their defining properties, and the applications each is relevant to. Functional distinctions over textbook definitions.
A plain-language explanation of developmental biology as the science of how organisms form, grow, and repair. Why it matters for technology, medicine, and conservation, not just academic research.
Why Tech Bio Fails
Tools built for academic novelty rather than industrial workflows. Microfluidic systems too complex, expensive, slow. How to test whether your product fits real demand.
Research-grade reagents cannot enter clinical pipelines. GMP, documentation, traceability requirements. The gap between interesting lab tool and regulated product.
Imaging, culture, data, and automation systems built in isolation. Manual workflows dominate. Why poor integration limits adoption beyond expert labs.
Cells lose viability and function after freeze-thaw. Batch variability increases with storage. Critical for clinical trials and commercial supply chains.
Current assays miss subtle stem cell state shifts. Epigenetic instability, differentiation drift, and what this means for regulatory submissions and clinical outcomes.
The physics and biology of scale-up: shear stress, nutrient gradients, contamination. Where suspension culture and bioreactor platforms lose cell quality.
How batch-to-batch variability and operator-dependent outcomes block commercialisation. Why pharma partners walk away from inconsistent data.
Second pillar series overview examining systemic reasons TechBio ancillary technologies fail in developmental and stem cell science.
Positioning Tech Bio
How established knowledge in laboratory and domesticated species (mouse, sheep, cattle, pig, human) can inform product development for less-explored applications.
How to describe what your product does in terms that resonate with biologists and engineers who work in developmental and stem cell science.
How to identify and reach buyer decision makers across disciplines trained as an engineer, physicist, developmental or stem cell biologist or business person.
How to test whether the scientists and engineers you think need your tool actually do. Methods for validating demand before committing to product development.
When your category barely exists, how to assess who else operates in your space and differentiate on substance rather than marketing claims.
How to map which specific workflows and bottlenecks your product addresses. Identifying the right application context before defining buyers.
Pillar article for third blog series on how TechBio companies should approach positioning their product in developmental and stem cell science.
Species preservation
How established knowledge in laboratory and domesticated species (mouse, sheep, cattle, pig, human) can inform product development for less-explored applications.
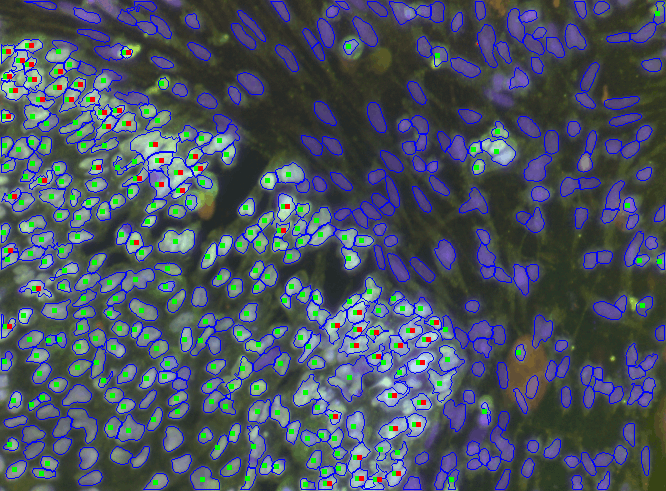


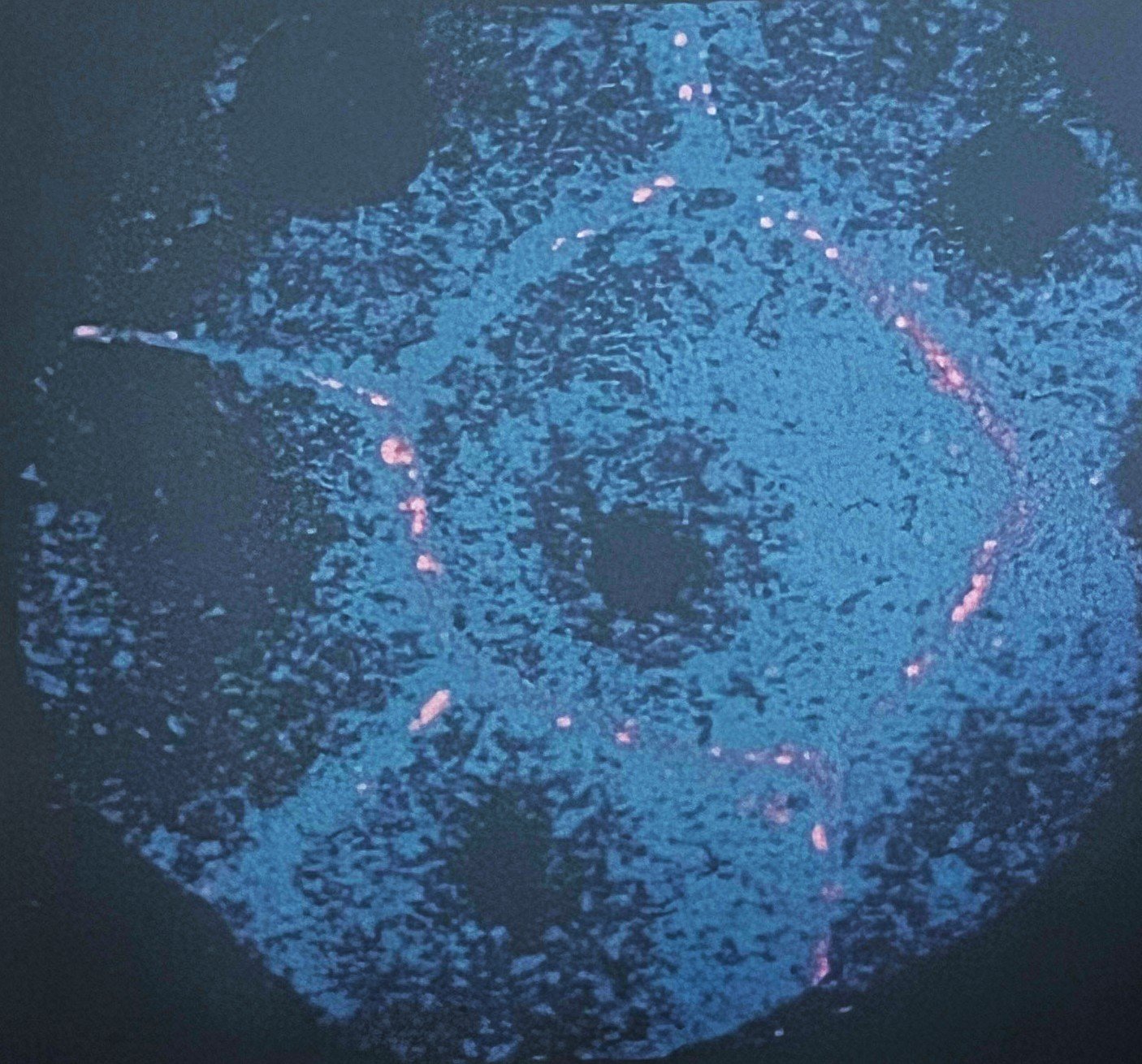




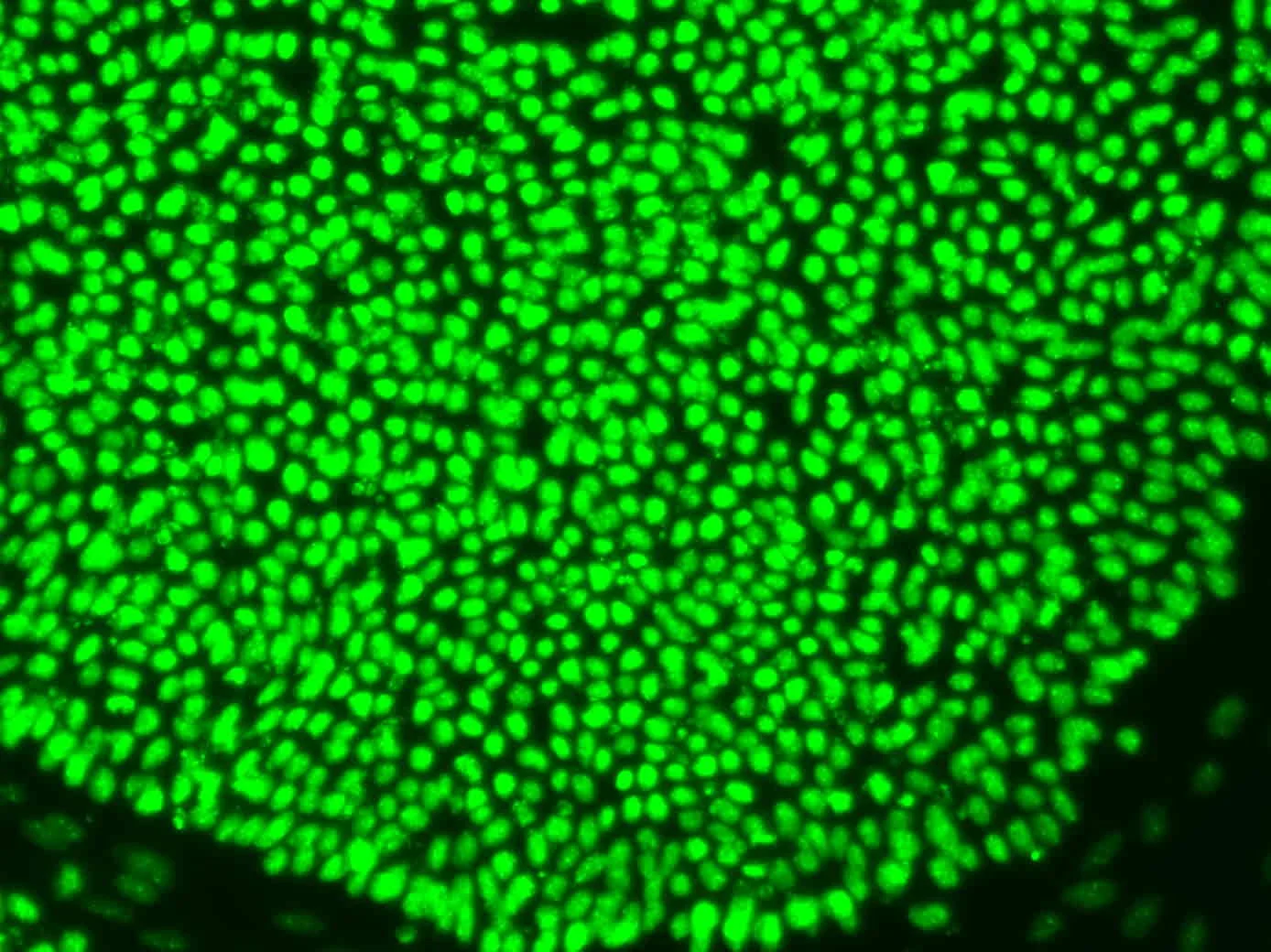
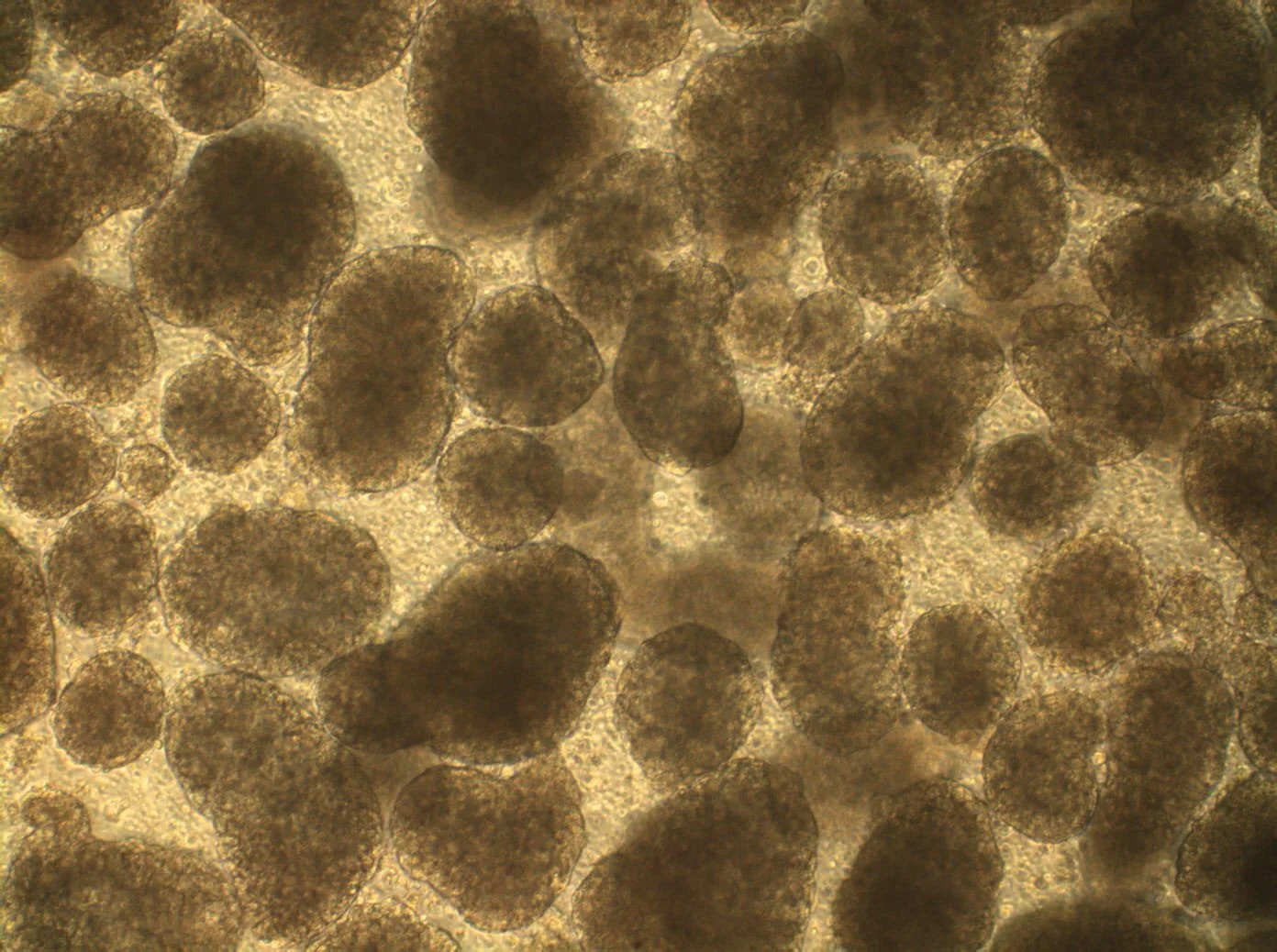
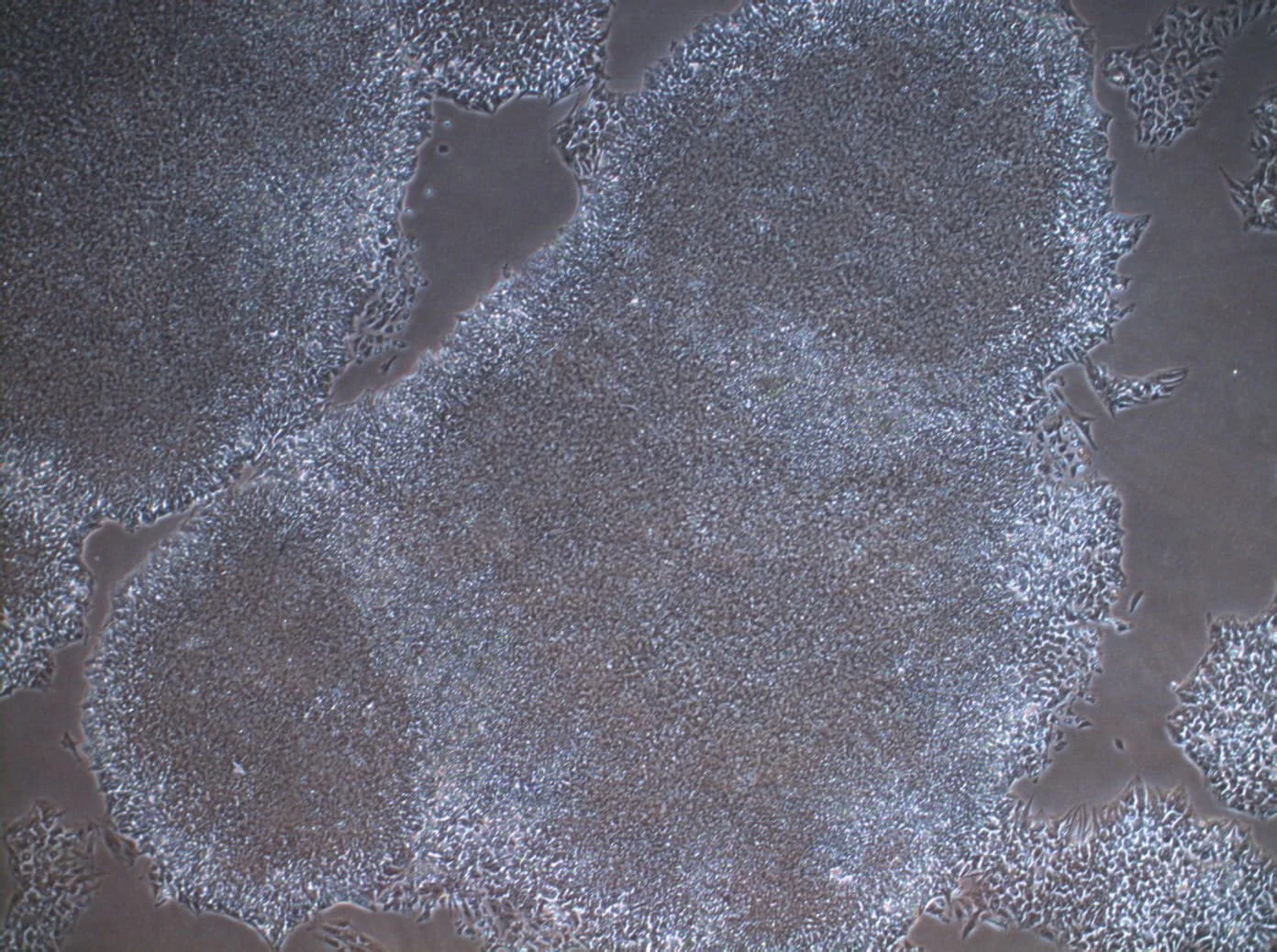
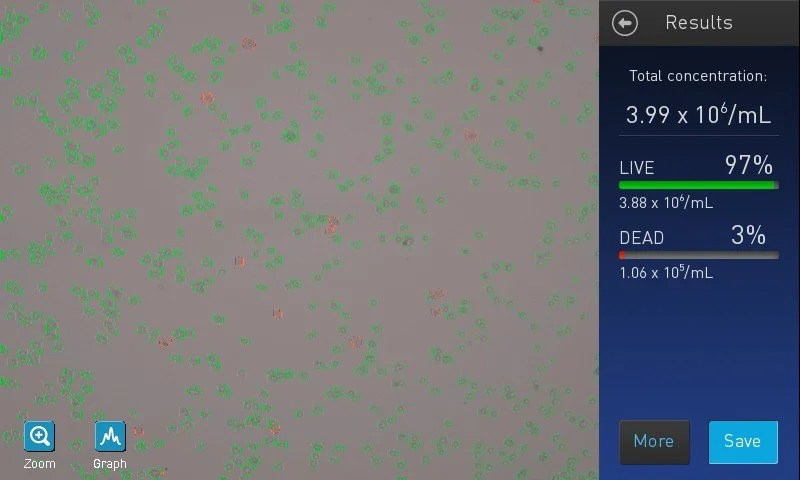






Introduces Paul de Sousa´s take on sustainability framework for the future of developmental and stem cell technology that runs through all of StemCells.Help blog series.