
Product-market fit in stem cell TechBio: building for workflows, not for papers
Tools built for academic novelty rather than industrial workflows. Microfluidic systems too complex, expensive, slow. How to test whether your product fits real demand.

The regulatory cliff: moving from research-grade to GMP-compliant product
Research-grade reagents cannot enter clinical pipelines. GMP, documentation, traceability requirements. The gap between interesting lab tool and regulated product.

Toolchain fragmentation: why imaging, culture, and data systems need to talk to each other
Imaging, culture, data, and automation systems built in isolation. Manual workflows dominate. Why poor integration limits adoption beyond expert labs.

Cryopreservation and supply chain fragility in clinical-grade cell products
Cells lose viability and function after freeze-thaw. Batch variability increases with storage. Critical for clinical trials and commercial supply chains.
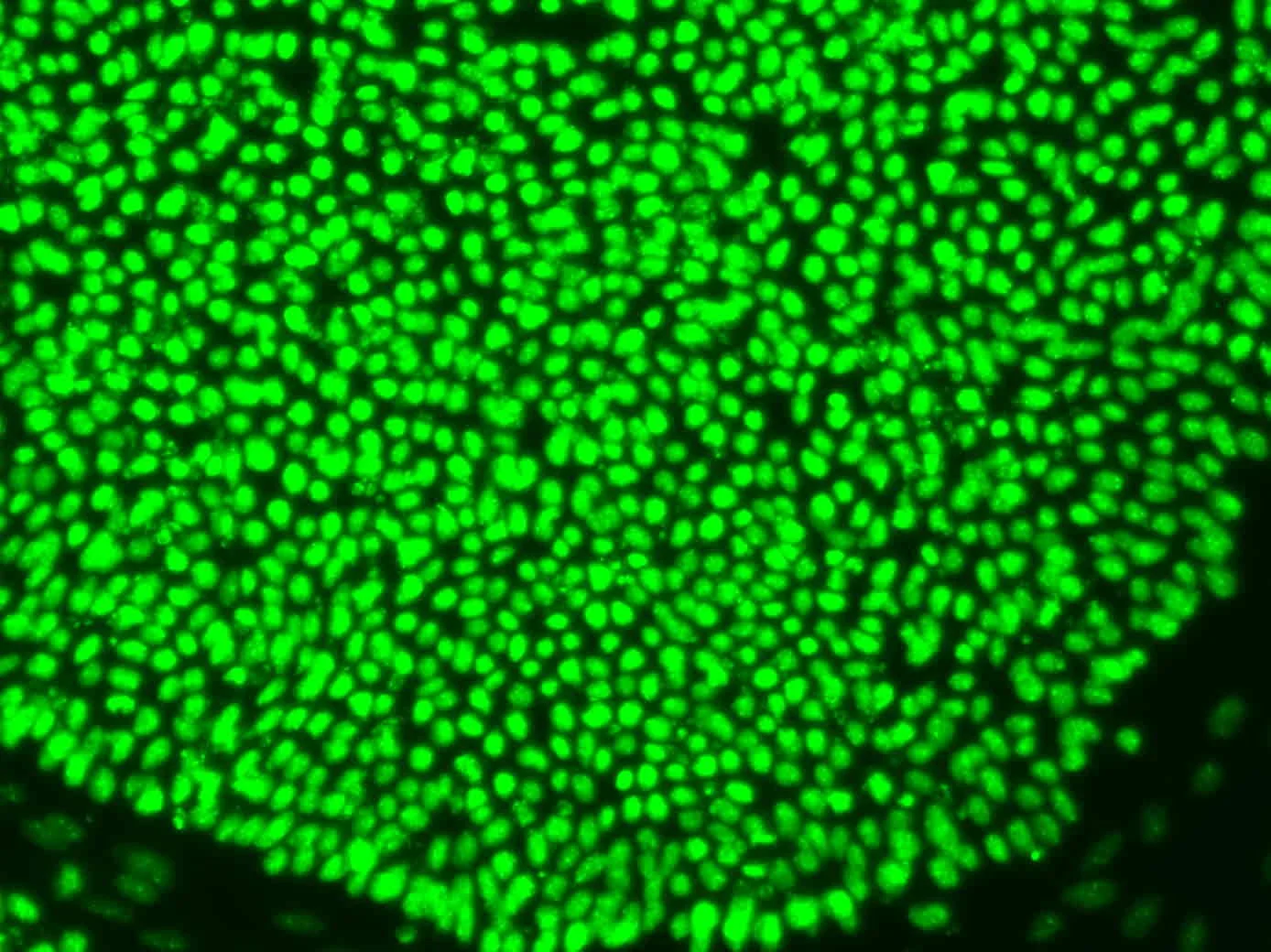
Characterisation gaps: what current assays miss about stem cell quality
Current assays miss subtle stem cell state shifts. Epigenetic instability, differentiation drift, and what this means for regulatory submissions and clinical outcomes.
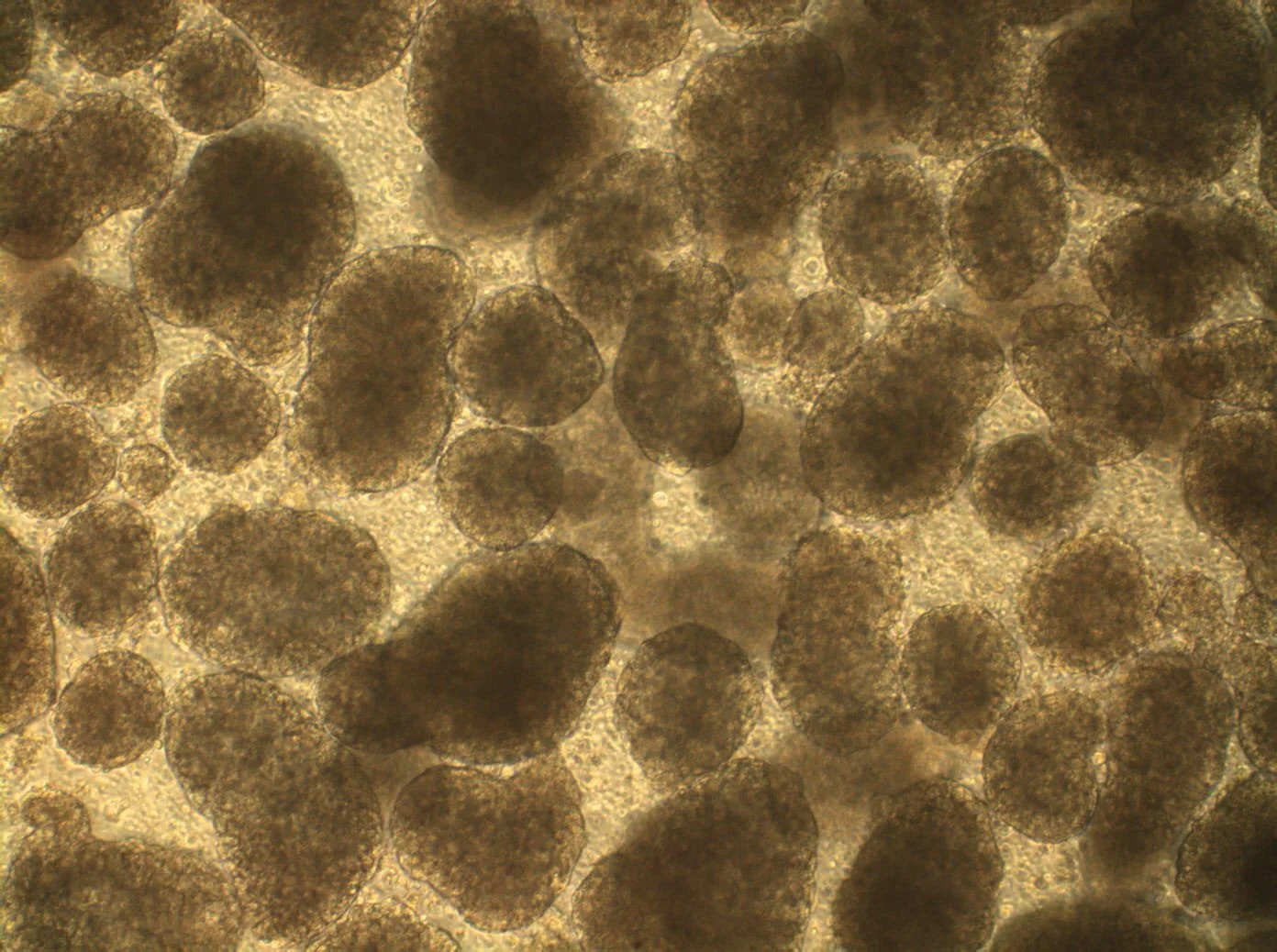
When lab-scale biology meets manufacturing reality: where most scale-up goes wrong
The physics and biology of scale-up: shear stress, nutrient gradients, contamination. Where suspension culture and bioreactor platforms lose cell quality.
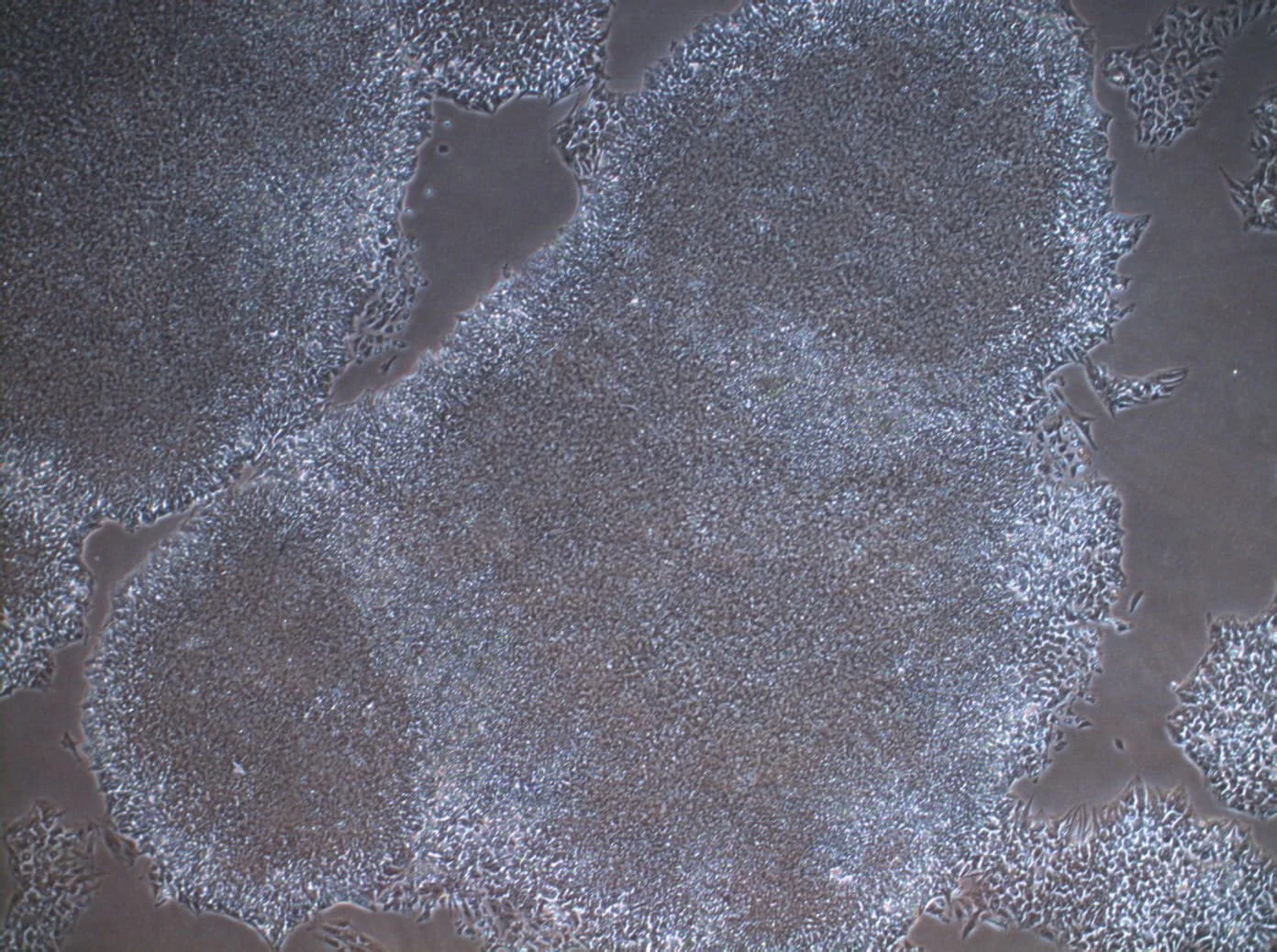
Reproducibility as a commercial problem, not just a scientific one
How batch-to-batch variability and operator-dependent outcomes block commercialisation. Why pharma partners walk away from inconsistent data.
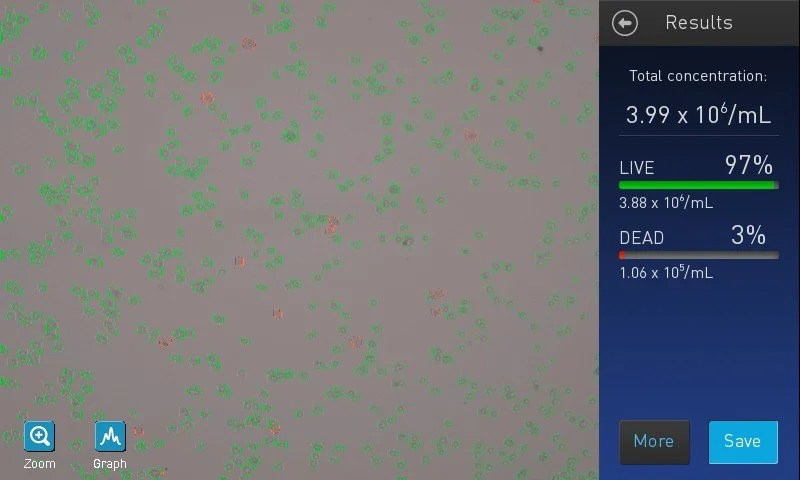
Why ancillary technologies for stem cell science fail, and what to do about it
Second pillar series overview examining systemic reasons TechBio ancillary technologies fail in developmental and stem cell science.